An ion ˈ aɪ ɒ n-ən is an atom or molecule with a net electrical charge. We wont ask you to name all 118 known elements.

Ib Chemistry Ellesmere College 4 1 Ionic Bonding And Structure
In 2016 nihonium Moscovium Tennessine and oganession were officially named and added to the periodic table.

. Considering some elements have had more than one name throughout history that would be a. Everyone knows helium hydrogen copper and gold. However have you kept up with the newest elements.
This transfer of electrons is termed electrovalence in contrast. Ionic bonding is a type of chemical bond that involves the electrostatic attraction between oppositely charged ions and is the primary interaction occurring in ionic compoundsThe ions are atoms that have lost one or more electrons termed cations and atoms that have gained one or more electrons termed anions. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton which is considered to be positive by conventionThe net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons.

Vektor Stok Crystal Lattice Structure Ionic Compounds Ionic Tanpa Royalti 1926335822 Shutterstock
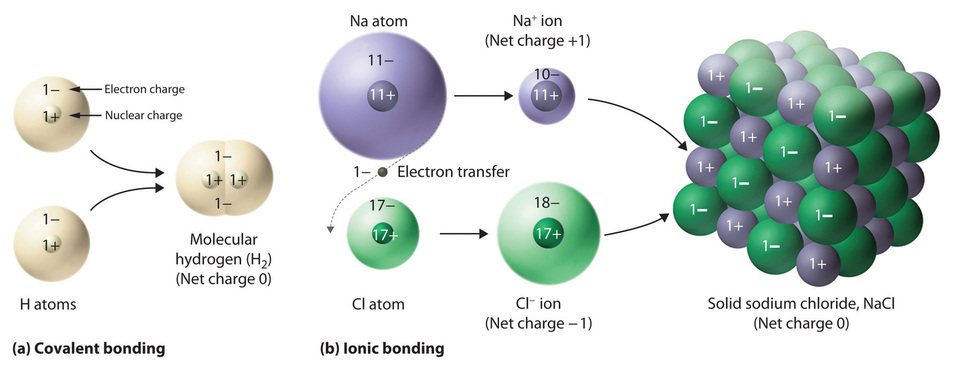
2 11 Ions And Ionic Bonds Chemistry Libretexts

What Do We Call A Molecule Of An Ionic Compound Since They Are Not Molecules Actually Can We Say One Molecule Of Sodium Chloride Nacl Quora
0 Comments